More than 30 people in different states of US reported side effects after having the shot with symptoms ranging from dizziness, nausea and fainting…reports Asian Lite News
Several states in the United States have temporarily shut down some vaccine sites after people suffered adverse reactions to the shot.
A total of 18 people in North Carolina reported side effects, while 11 people in Colorado reacted to the shot with symptoms ranging from dizziness, nausea and fainting, according to media reports, Xinhua news agency reported on Monday.
Georgia was the third US state to temporarily shut down a vaccine site following North Carolina and Colorado, after eight people suffered adverse reactions following vaccination.
According to local officials, one person was sent to a nearby hospital for evaluation before being released. The other seven were sent home after being monitored at the site.
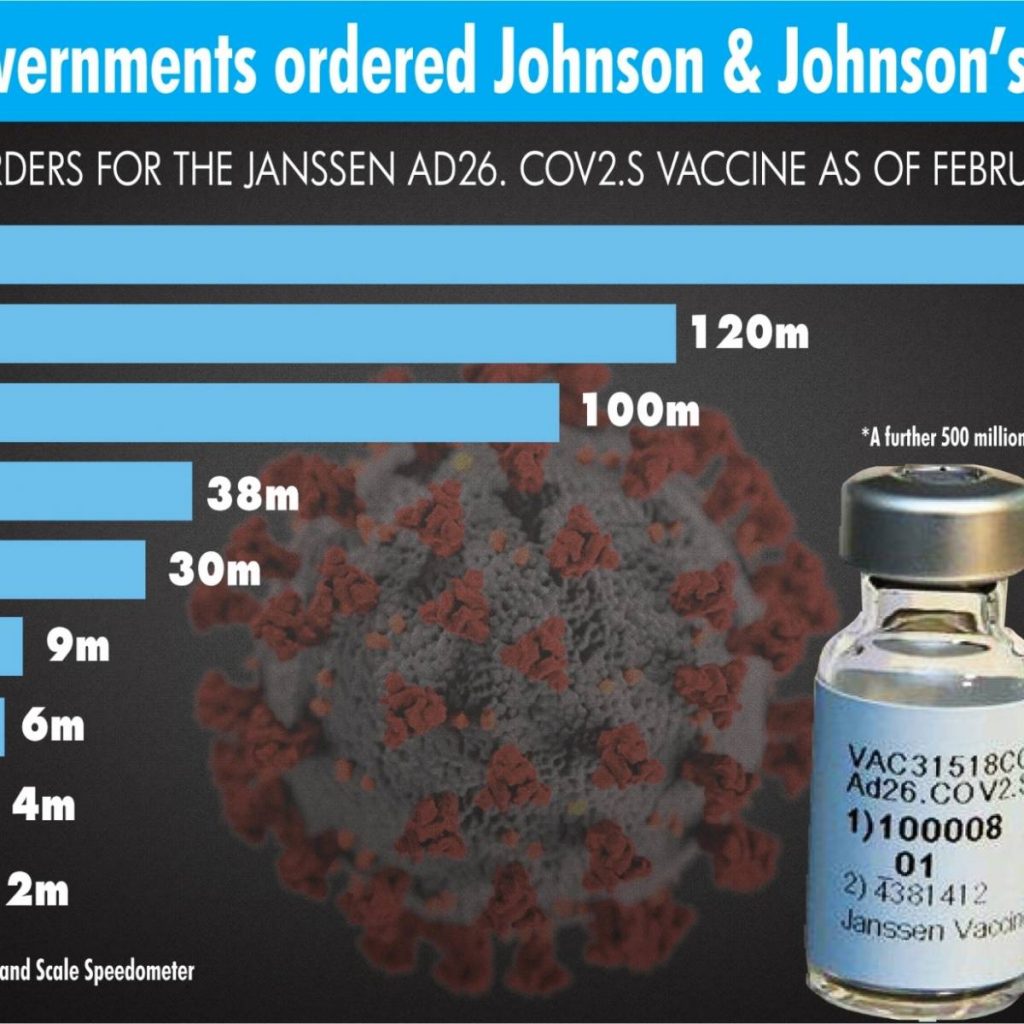
The US Centers for Disease Control and Prevention (CDC) said its analysis “did not find any safety issues or reason for concern.” The agency recommended that healthcare providers continue to administer the Johnson & Johnson vaccine.
Another problem facing Johnson & Johnson is vaccine distribution. The supply of Johnson & Johnson’s Covid-19 vaccine is expected to drop by 85 per cent nationwide this week, after the company suffered production issues at a facility in Baltimore, Maryland, according to federal officials and data.
Also read:World Leaders Unite For Int’l Pandemic Treaty
A manufacturing subcontractor in Baltimore mixed ingredients from the Covid-19 vaccines of Johnson & Johnson and AstraZeneca, delaying shipments of the Johnson & Johnson shot across the nation.
The plant is run by Emergent BioSolutions, a manufacturing partner to both Johnson & Johnson and AstraZeneca. Federal officials attributed the mistake to human error, which resulted in contamination of up to 15 million doses of Johnson & Johnson’s vaccine.

Jeff Zients, the White House coronavirus coordinator, said Friday that the company is still working to address the production issues in Emergent BioSolutions, which is not certified yet by the US Food and Drug Administration (FDA).
Johnson & Johnson’s Covid-19 vaccine was approved for emergency use in the United States in late February. It is the third Covid-19 vaccine that has received FDA’s authorization, also the first single dose Covid-19 vaccine available in the United States.
As of Monday, about 189 million Covid-19 vaccine shots have been administered, while more than 237 million Covid-19 vaccine doses have been distributed across the country, according to data of the CDC.
Also read:India donates 100,000 doses of Covid vaccine to Nigeria